There were challenges in the Levetiracetam sales in the past few quarters, which is the biggest revenue driver in prime segment of GDS for Neuland. Management reasoned that it was because of lower customer demand & customer holding inventory. But it hasn’t changed even after 3 quarters. In yesterday’s earnings call also, management said the same commentary.
(You can hear the discussion about Levetiracetam sales from 34:13 in Q1FY23 call)
I’m adding past earnings call snippets to understand the progression of this issue and how management justified the issue.
Q3 FY21: Levetiracetam & Mirtazepine are the key growth drivers in GDS. Will continue to invest in those molecules and become market leaders.
Q4 FY21: Our GDS business was led by key molecules Levetiracetam & Mirtazepine.
Q1 FY22: We have gained market share and increased margins on Levetiracetam & Mirtazepine.
Q2 FY22: For Levetiracetam, we’re strong in the markets that we’re in but we’re NOT seeing very strong spurt over there.
Reason- these are also the CYCLES that you have for these products.
Q3FY22: Products which were historically doing well like Levetiracetam & Mirtazepine didn’t perform to our expectations.
Reason- Lower customer off take & customer holding on to inventory from the orders in the past or they don’t have a production campaign or need right now.
Q4FY22: Levetiracetam which has been doing well for us historically had challenges this year.
Reason- We believe that this is because of low customer off take, perhaps because of high inventories and we expect that the volumes should recover in coming quarters.
Yesterday’s con call
Q1 FY23: Biggest impact in the GDS was from Levetirazetam. It is also the largest product in our GDS segment which impacted our performance. We think it is temporary.
Reason- because of combination of reduction in demand & inventory build up at customer’s end.
As Mr.Aditya Khemka pointed out in the earnings call, if it was due to inventory build up at customer’s end, it would have been temporary. If the challenge is due to increased competition from peers, then it is alarming.
For Neuland the issues with Levetiracetam API sales started from Sept-Oct 2021 and it is persisting even now.
Another interesting thing to note here is that on May 2021, Divi’s has talked about expanding their Levetiracetam manufacturing with improved changes (background integration, increasing yield, solvent recovery, conserving raw materials) and the expansion to be completed within 3 months. That is the time period where gradually challenges arose in Levetiracetam business for Neuland.
Neuland management never acknowledged increasing competition in Levetiracetam sales or losing market share for their biggest revenue driver in generic business. They always had same explanation of “lower customer off take & customer holding inventories”. They could have given more clarity on this situation.
CMS business is also stagnating for so many quarters. I’ve written detailed post about these in the past.
Even after having 21 molecules in the commercial CMS stage, their CMS commercial revenues are not showing significant growth. I’ve talked about how Austedo (Deutetrabenazine) revenues are growing for Teva in clear contrast to Neuland’s CMS revenue growth.
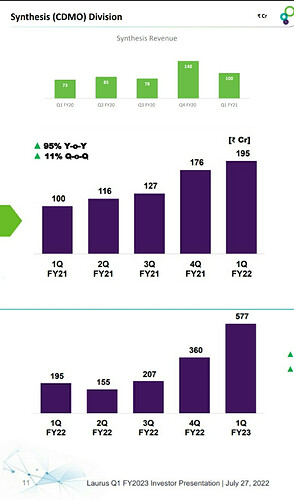
For a different perspective, this is how Laurus scaled up their custom synthesis in the last 3 years.
I don’t know if it is fair to compare custom synthesis of Laurus to that of Neuland since molecules and customers are different. But lack of momentum or lack of growth is pretty much visible in the case of Neuland. In the yesterday’s earnings call, management has said the reason for the weak Custom synthesis was because of technical challenges they faced during scaling up a molecule from lab to commercial scale and subsequent delay in delivering molecule to the innovator. This is something an innovator doesn’t seek in their manufacturing partner. Innovators always want timely delivery from their manufacturing partner and this reason can compel them to give contract to someone else.
| Subscribe To Our Free Newsletter |