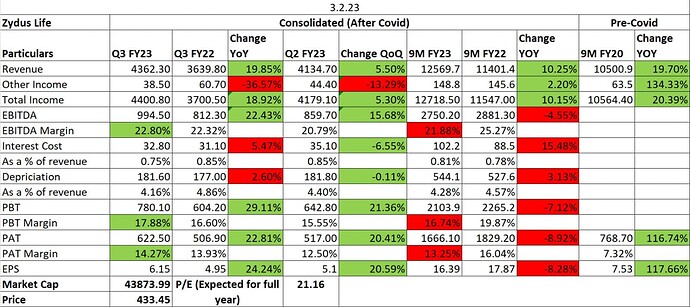
- EBITDA Margin guidance of 20% is maintained by management which is consistent with their guidance in Q2 FY23.
- They said in Q2FY23 that they are planning 25-30 launches in US in FY23 which they are consistent with. They filled 6 ANDA’s and 14 ANDA approvals they have got. Launched 6 new products as well. US Business is expected to grow in the next 2-3 years.
- Expected spend on R&D is 7-8% of revenues is what they said in Q2 FY23 which is consistent.
- Revlimid will still see high sales in Q4 as seen in Q2/Q3. There will be 4 transdermal launches in the next few years.
| Subscribe To Our Free Newsletter |