BATTERY
The Inflation Reduction Act specifies that, starting in 2024, no EV that contains battery components produced by a foreign entity of concern18—defined as China, Russia, Iran, and North Korea—will be eligible for the credit. Beginning in 2025, the same becomes true for critical minerals used in EV batteries.
Now how did I confirm this? AMI Concall
:

Here is as per the chemistry – company wise :
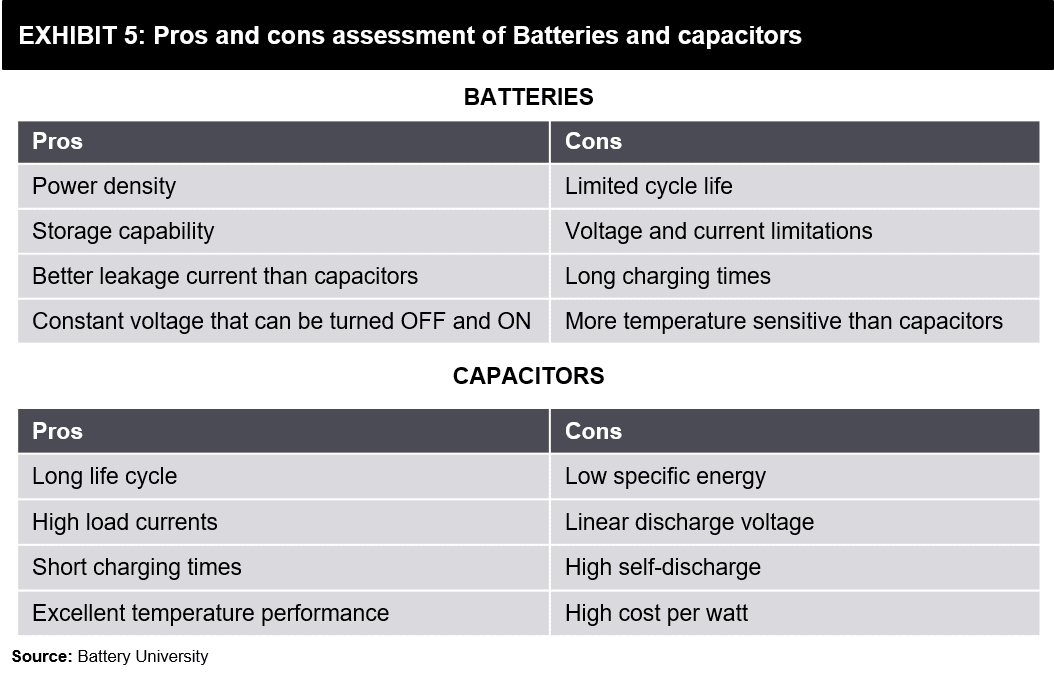
- Key Ingredient – Tata Chemical (Soda Ash)
- HSCL : Lithium Ion Battery components
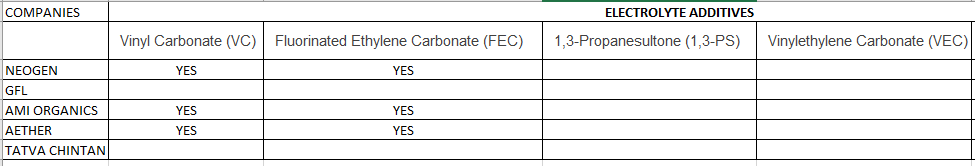
- Electrolyte Salts – GFL, Neogen, Tatva Chintan
- Electrolyte Additive – AMI Organics, Aether Industries
- Solvent/Catalysis Profile : Tatva Chintan, Balaji Amines
Electrolyte Additive types
- Vinyl Carbonate (VC)
- Fluorinated Ethylene Carbonate (FEC)
- 1,3-Propanesultone (1,3-PS)
- Vinylethylene Carbonate (VEC)
- Others
Solvents
- Ethylene Carbonate (EC): It’s commonly used as a solvent for the electrolyte in lithium-ion batteries due to its high dielectric constant and ability to dissolve lithium salts.
- Dimethyl Carbonate (DMC): Often used as a co-solvent in combination with other solvents like EC due to its good solubility with lithium salts. (Balaji Amines, Tatva Chintan, Paushak)
- Diethyl Carbonate (DEC): Another solvent used in combination with EC and DMC in the electrolyte of lithium-ion batteries. (Tatva Chintan)
- Ethyl Methyl Carbonate (EMC): Similar to DEC, EMC is used as a co-solvent to improve the electrolyte’s properties.
- Propylene Carbonate (PC): It’s also a common solvent used in lithium-ion battery electrolytes due to its high dielectric constant and ability to dissolve lithium salts. (Balaji Amines)
- Gamma-Butyrolactone (GBL): This solvent can be used in sodium-ion battery electrolytes due to its ability to dissolve sodium salts and improve ionic conductivity.
- 1,2-Dimethoxyethane (DME): It’s another solvent occasionally used in sodium-ion battery electrolytes to enhance the performance of the system.
- Sulfolane: This high boiling point solvent is sometimes used in sodium-ion battery electrolytes due to its good stability and compatibility with sodium salts.
- Monoglyme is one of those solvents used as a mix combination with dimethyl carbonate or other solvents to dissolve the electrolyte salts. So, this becomes the
part of the electrolyte solution within the Lithium battery (Tatva Chintan)
Now Electrolyte Additive can be used either in Sodium Ion or in Lithium Ion Profiles.
The real threat is to the Electrolyte salts as in case if market changes to go for Sodium Ion:
Here are their common variances :
- Sodium-ion batteries might use sodium salts like sodium hexafluorophosphate (NaPF6), sodium chloride (NaCl), sodium perchlorate (NaClO4), or other sodium-based compounds.
- Lithium-ion batteries commonly use lithium salts such as lithium hexafluorophosphate (LiPF6), lithium carbonate (Li2CO3), lithium perchlorate (LiClO4), and lithium tetrafluoroborate (LiBF4).
Lithium – We do know that companies like Neogen, GFL, Reliance are making a mark.
For Sodium ion – only 4-5 companies have developed this product and has patent.
- Faradian (Acquired by Reliance)
- BYD (China)
- KPIT (India)
- CATL : Cherry Auto (Chinese)
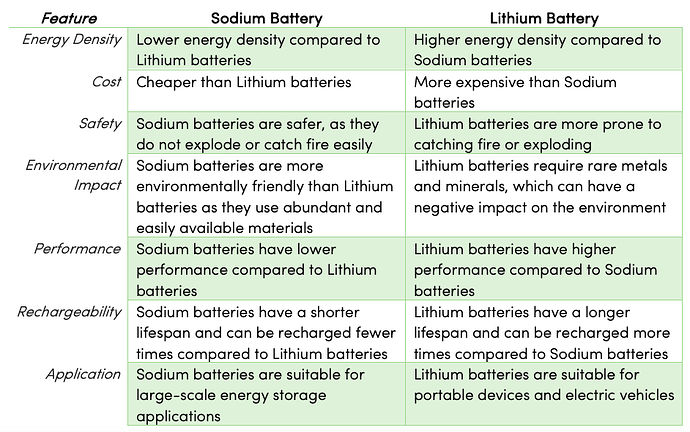
Coming to the profile of Sodium vs Lithium comparative difference
Credits : Lithium-ion vs. Sodium-ion Batteries: Sustainable Energy Options for the Future
Since i spoken about batteries why not Super capacitors than only the battery side we are looking into?
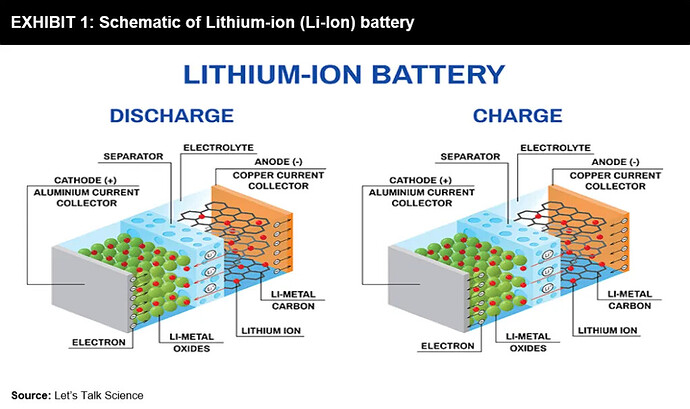
Lithium Ion
- Relies on Chemistry reactions
- Consist of Anode and Cathode
How it works : These two sides are submerged in a liquid electrolyte and separated by a micro-perforated separator, allowing only ions to pass through. During batteries’ charging and discharging, the ions tend to flow back-and-forth between the anode and cathode. While this ion transfer process occurs, the battery gets heated up, expands, and then contracts. These reactions gradually degrade a battery, resulting in a reduced lifespan of batteries.
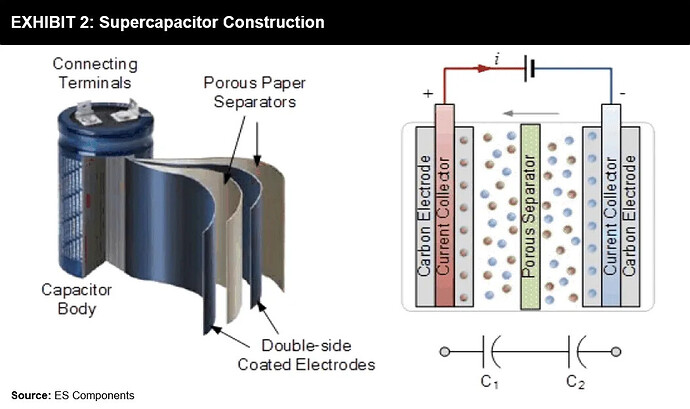
Super Capacitors
- Don’t rely on Chemical Reactions
- They store potential energy electrostatically within them
Supercapacitors use dielectric or insulator between their plates to separate the collection of positive (+ve) and negative (-ve) charges building on each side’s plates. It is this separation that allows the device to store energy and quickly release it. It basically captures static electricity for future use.
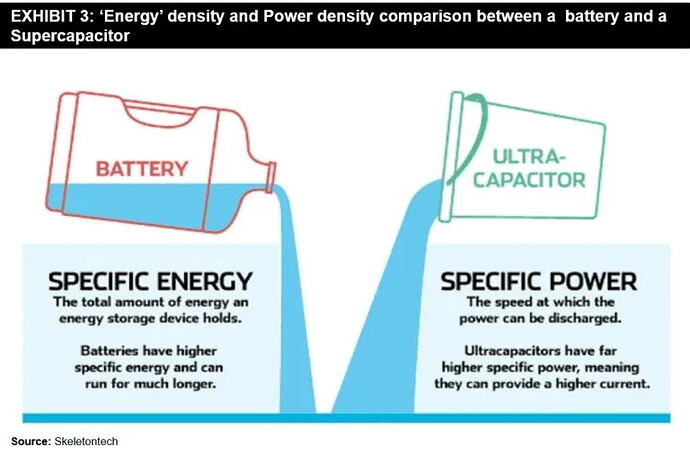
Super capacitors are good for immediate burst of power.
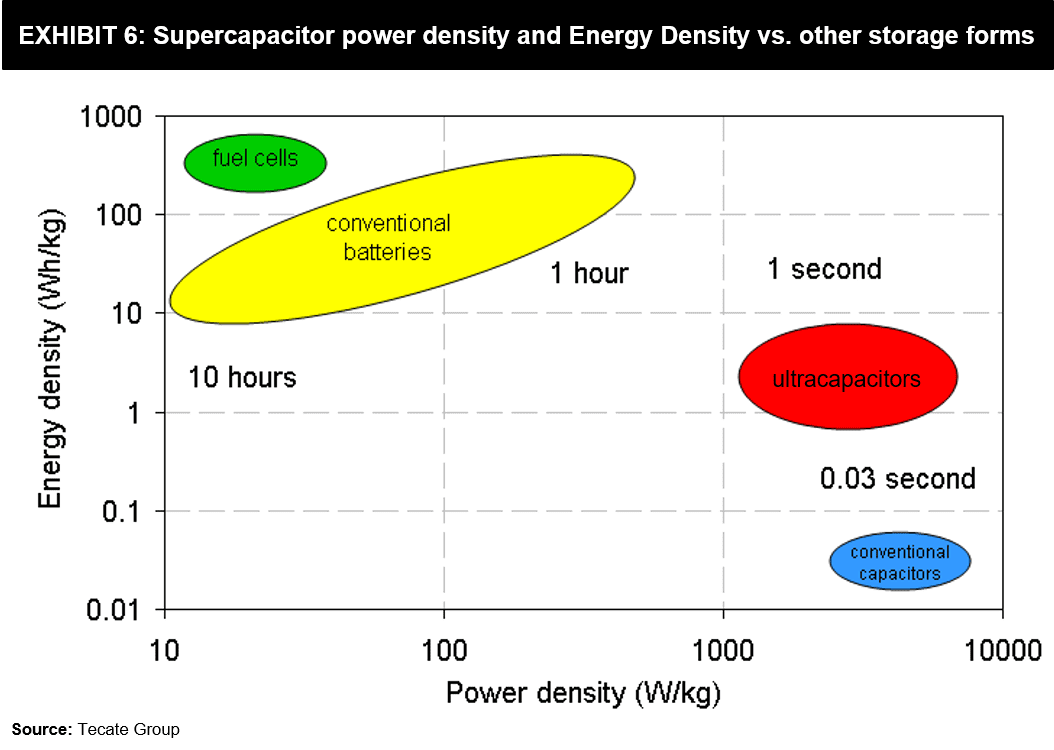
Power Density, Energy Density vs Storage Forms
Credits : Supercapacitors – A Viable Alternative to Lithium-Ion Battery Technology? – FutureBridge .
Going forward now they are trying to collaborate Lithium + Supercapacitors = naming it Lithium Ion Capacitors
Here is the video : https://www.youtube.com/watch?v=XlON0Uj2Zk0
Coming to the Indian company profiles
Capex Plans – All of them have plans to expand based on their chemistry.
Environment Clearance
- Tatva Chintan : https://environmentclearance.nic.in/DownloadPfdFile.aspx?FileName=fgftrmkwVeXyTcQelv2gWFJL3QzFq5qLxOqYwfPDYCdMNPl6td6l3jIDNGgvnquPj6Tkbklk7Qu3XpwsBEeRVW374LqDQYtnIDU3EB6tyQq3C2hsR8Nh4wR4YO00qXR4ep/9bER2jqKROPPHJ2GdYZjuLMN6VT7NAyqLyRP4eU0WaRkDA8Vp5qmlPa7uSY67&FilePath=93ZZBm8LWEXfg+HAlQix2fE2t8z/pgnoBhDlYdZCxzUlDadBGu7t8v4JoQvNU6UBlSmL0YQ7WQYaxkvlQvexKQ==
- Balaji Amines : https://environmentclearance.nic.in/writereaddata/Online/TOR/0_0_13_Jul_2015_190624310RT2EHBriefSummary.pdf
| Subscribe To Our Free Newsletter |